What is quality system management software?
We’ve all seen this happen. A customer gets a faulty product because someone followed an old instruction manual buried in an email thread. At some point, most businesses hit the spreadsheet ceiling, where tracking work through scattered files stops being manageable and starts creating risk.
Basic quality control checks finished products for mistakes after they happen. Quality management is broader. It puts systems in place to stop those mistakes in the first place.
That is where quality management software, or QMS, comes in. A good QMS is a central system for how work should be done. It does not just store records. It helps teams follow the right process at the right time.
Business teams often talk about a single source of truth. In simple terms, that means one central place where the current, approved way of working lives. When you replace scattered document versions with dedicated quality software, you cut confusion fast. Everyone works from the same instructions, and that usually means better work and fewer do-overs.

From Filing Cabinets to Digital Brains: Defining the Core Components of a Modern eQMS
Good records matter, but paper systems get messy fast. An electronic Quality Management System, or eQMS, gives teams one digital place to manage quality work. It holds approved processes, tracks actions, and reminds people what needs attention.
Instead of leaning on manual spreadsheets, the software usually brings quality work into four main parts:
Document Control: One central place for current, approved instructions.
Training: Alerts and records that show who has read and understood new procedures.
Audits: Routine checks to confirm standards work in day-to-day practice.
CAPA: Corrective and Preventive Action tools that help find the root cause of a problem and fix it for good.
Tying these parts together is the audit trail. That is the system record of who did what and when. With automated CAPA workflows, the software handles much of the follow-up work. Reminders move tasks along, so managers do not have to chase every step by hand.
When teams digitize these steps, miscommunication drops and the work becomes more consistent. Every part matters, but document control is usually where that consistency starts, whether you are in a regulated industry or just trying to run a growing business with less confusion.
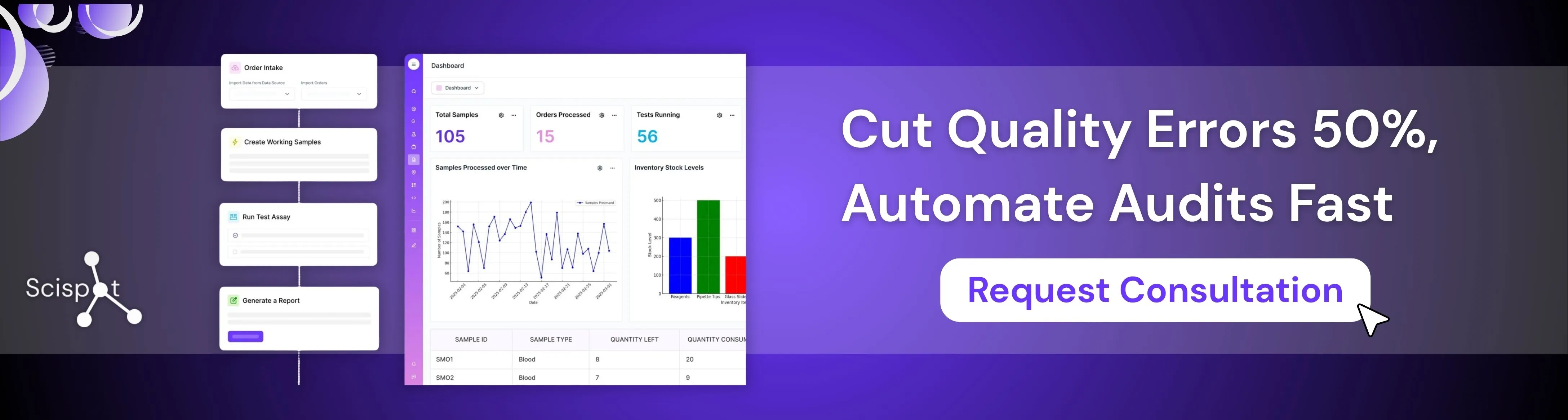
Scispot as a Smarter Quality Management Solution
Scispot is a strong fit for quality system management because it brings document control, training, approvals, audit trails, and corrective actions into one connected system instead of splitting them across email, spreadsheets, and separate tools. Teams can manage SOPs with version control, route reviews with secure electronic signatures, track changes with full traceability, and automate follow-ups so quality work does not stall.
For growing labs and regulated teams, Scispot also makes it easier to stay inspection-ready. It combines day-to-day usability with workflows built for compliance, which helps teams cut manual work and build a more consistent quality process.
The Document Control Secret: Why the “Latest Version” Is Your Most Powerful Tool
Old instruction manuals have a way of coming back. Someone finds one in an email thread, uses it by mistake, and the team pays for it later. That is a real operational risk.
Version control fixes this by making one system the source of truth. Old drafts are archived, and teams see the newest approved version. With dedicated quality software, the odds of someone following last year’s process drop sharply.
Moving approvals into a digital system also changes how teams handle reviews. Instead of tracking down a manager for a signature, modern platforms use secure electronic signatures and route the document to the next reviewer automatically. That keeps documents moving and creates a clear, time-stamped record of who approved what.
At first, relying on digital approvals can sound risky, especially in regulated industries. But these signatures are built to function like legally binding handwritten signatures and can support FDA 21 CFR Part 11 requirements, which are meant to keep electronic records authentic and tamper-resistant.
Closing the Loop: Using CAPA and Non-Conformance Tools to Solve Problems Forever
Even with solid instructions, mistakes still happen. A wrong part ships. A product fails a test. In quality management, that is a non-conformance. Good software captures those issues instead of letting them disappear. Customer complaints and internal failures become signals the business can use to improve.
That is also why non-conformance management software can pay for itself. Preventing even one major recall can cover the cost of the system.
Still, fixing the immediate issue is not enough. You need a closed-loop process that lowers the chance of the same problem happening again. That is where CAPA tools help. They guide teams through root cause analysis using methods like the 5 Whys:
- Log the symptom: Record the exact issue in the central system.
- Ask why: Use required fields to move past the surface error and find the broken process.
- Assign the fix: Route the task to the right person so the procedure gets updated properly.
A structured process keeps teams from solving the same problem again and again. The software can also check whether the fix worked before the case is closed.

Industry Superpowers: Tailoring Quality Tools for Pharma, Automotive, and Construction
Every business needs to catch mistakes, but the rules and day-to-day risks vary by industry.
Pharma companies use quality management software to support GxP compliance, which covers strict rules meant to keep medicines safe. These systems often require people to sign off on each step before work moves forward.
Automotive teams care deeply about traceability and fast error detection. Quality management software in that setting helps managers watch the shop floor in real time. If a machine starts placing a part incorrectly, the system can flag it before defective cars leave the line.
Construction has a different problem. A lot of the work depends on outside vendors and contractors. That is why construction quality software often focuses on supplier communication. Teams use supplier portals to confirm that materials meet requirements before they ever arrive on site.
Whether a company is making cough syrup or pouring concrete, the software has to fit the work. But one thing stays the same. These tools only work well when everyone can get to the same information without delay.
Why the Cloud Wins: The Benefits of Transitioning to an Electronic QMS
Few things are more frustrating than needing a key file that sits on one office computer. Cloud software removes that problem. With Software as a Service, or SaaS, teams access the platform online while the provider handles storage, updates, and much of the security work.
One of the biggest benefits of moving to an electronic QMS is simple. People can review and approve work from anywhere with a secure internet connection.
Compared with local servers, cloud-based quality systems usually offer four main advantages:
- Anywhere access: Teams can review and approve documents on the go.
- Less IT burden: The provider handles much of the security and maintenance work.
- Automatic upgrades: New features and compliance updates roll out without manual installs.
- Lower hardware costs: Companies avoid the cost of maintaining on-site infrastructure.
Certifications used to trigger a once-a-year scramble through paper files. Cloud systems change that. With tools built for standards like ISO 9001, businesses can stay audit-ready on an ongoing basis because records stay current and organized.

Getting Your Team on Board: How to Implement New Quality Workflows Without the Headaches
Even the best software fails if people do not use it. When rolling out new quality workflows, it helps to focus on people first and software second.
Instead of changing everything at once, start small. A soft launch with a test group works better than a company-wide switch overnight. One early win, like five employees approving a document successfully in the new system, can build trust without overwhelming the team.
As that group gets comfortable, training records become more important. A training matrix makes it clear who is qualified for what. If someone missed a required safety course or has not reviewed an updated SOP, the system can flag it right away.
Adoption grows when teams see real progress. In the first month, it helps to aim for a few practical wins, such as cutting approval time in half or running one review process without paper. Those steps also make audits easier because records are already in place and linked to trained staff.
Once people trust the system and use it daily, the shift becomes much easier. That is when teams move from reactive work to a more stable way of operating.
From Chaos to Confidence: Your Path to Operational Excellence
Quality work does not need to feel like a hunt through messy spreadsheets and old files. When teams move away from scattered paperwork, they gain something simple but valuable: clarity. The right process is easier to find, easier to follow, and easier to prove.
A practical way to start is with a short first-30-days plan:
- Review your current manual processes and find where time is being lost.
- Compare quality platforms, but judge them first on ease of use, not just feature count.
- Pick one small workflow to automate so the value is easy to see.
One rule helps through the whole process. If the system makes daily work harder, it is the wrong system. The right one should make work clearer, not heavier. Start with one workflow, make it work well, and build from there.
.webp)




.webp)
.png)
.webp)
























.webp)
.webp)



