How do bioanalytical labs ensure accuracy in testing?
A doctor setting the dose for a life-saving drug is not guessing. They rely on measurements that must be right down to a billionth of a gram. Under strict international safety standards, that level of accuracy can mark the line between an effective treatment and a serious complication.
Standard clinical blood work may check something like cholesterol. Bioanalytical testing, which measures complex drugs in biological systems, deals with a much harder problem. Scientists do not accept a raw number from a machine at face value. They need a validated result, backed by checks that show the data is trustworthy before it reaches a patient’s chart.
Keeping bioanalytical testing accurate takes more than expensive instruments. Human samples are messy and complex, so the lab’s job is to make sure each final number reflects what is really there. That level of accuracy comes from strict protocols that help keep modern medicine safe.

Why labs need both accuracy and precision
When doctors prescribe life-saving medicine, they trust that lab numbers are true. A simple way to think about bioanalytical lab accuracy is a dartboard. Accuracy means hitting the bullseye, finding the exact amount of a substance. Precision means the darts land in the same spot each time, even if that spot is off to the side.
That tight grouping, repeated over and over, is reproducibility. In regulated bioanalytical studies, reproducibility matters because a test that gives a different answer every hour is not useful. But consistency alone is not enough if the result is wrong every time.
Think of a bathroom scale that always adds two pounds. It is precise, but it has built-in bias. Managing precision and bias in clinical sample analysis means technicians keep adjusting and checking their tools so those steady results also point to the right answer.
Accuracy is a regulated requirement that helps keep medicine safe. Before labs test any human samples, they have to prove their methods actually work.
How labs prove their methods work before testing your samples
Before a single drop of patient blood is tested, the lab runs what is basically a full scientific rehearsal. Creating a new bioanalytical method is a bit like writing a recipe for a high-stakes bakery. A chef would not serve a wedding cake without a test bake, and scientists do not use a new method until they show it works under the conditions they expect. That step is called method validation.
Strict laws govern this process so medical data can be trusted. To meet FDA bioanalytical method validation guidelines, a lab has to show that the method measures what it says it measures. Evaluators check a set of quantitative performance traits to make sure the test will hold up when it matters.

To pass and be cleared for human use, the method has to show five things:
- Selectivity: It finds the right molecule, like pulling the right needle from a huge haystack.
- Sensitivity: It can detect very small amounts of a substance.
- Accuracy: It gives the true number.
- Precision: It gives the same result each time.
- Stability: The sample does not break down or change while it waits, even in storage.
Method validation protects patients from bad medical decisions. It is also hard to get right because biological samples are crowded and messy. Scientists have to separate background noise from the one drug signal they actually need.
Finding one specific drug in a sea of blood cells
Trying to find a drug in human blood is a bit like trying to hear a whisper in a stadium full of cheering people. Biological samples are full of proteins, fats, and other compounds that create background interference called a matrix effect. If that noise is not controlled, it can cause variability in pharmacokinetic results, the data that shows how the body processes a drug over time.
To deal with that, labs add a carefully measured reference chemical to the sample. This acts as an anchor, so the instrument can compare the unknown drug level against something known and stable. Internal standards are a key part of assay robustness because they help show the test is still working in a crowded biological environment. Minimizing matrix effects in mass spectrometry, the tool used to scan and weigh these molecules, helps make sure background noise does not hide the real signal.
Medical treatment depends on scientists winning this quiet fight against chemical noise every day. True accuracy means working through the natural mess of the human body to produce data that can be trusted. Good science helps, but strong oversight is what makes sure shortcuts do not creep in.

How rules like GLP protect data integrity
When labs test life-saving medicines, a promise to do good work is not enough. They follow strict international standards called Good Laboratory Practice, or GLP. You can think of GLP-compliant bioanalysis quality control as an audit for science. It tracks every step, every sample, and every action so the data stays honest, traceable, and protected from error.
Drug companies look for these standards when choosing a bioanalytical contract research organization, or CRO. Experts reviewing these labs usually watch for three red flags:
- No clear record of who handled a sample.
- Manual-only data entry, which opens the door to human error.
- Uncalibrated equipment, which can produce false readings.

Avoiding those risks is central to bioanalytical data integrity. The final result has to be complete, accurate, and unchanged. Modern labs support this with automated audit trails, which create permanent digital records of each action as it happens. These rules are not just paperwork. They help protect the quality of medical results. And to reduce the remaining risk in manual record-keeping, many labs now rely more on digital systems.
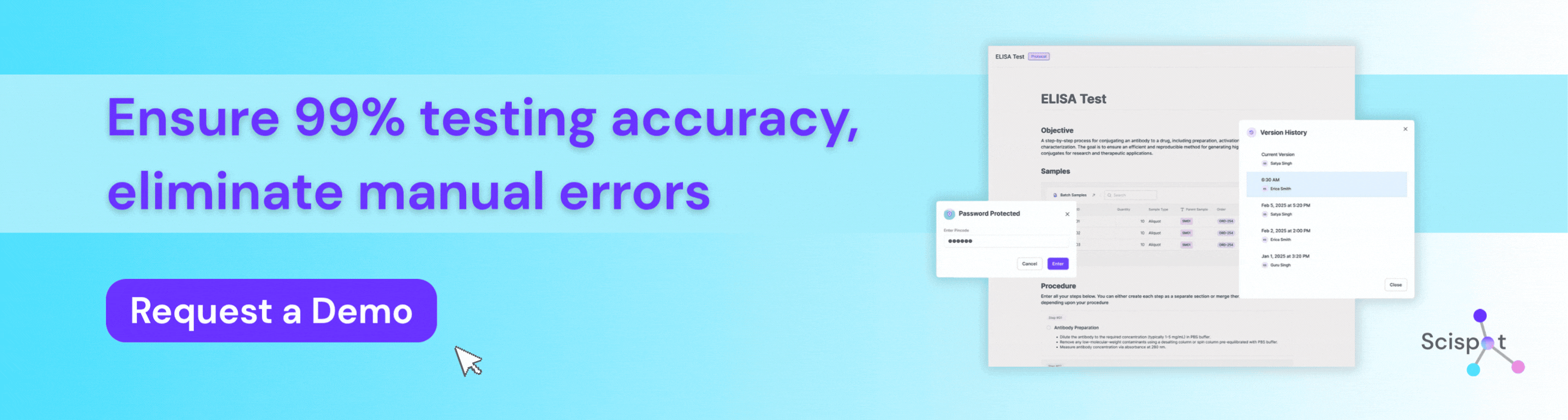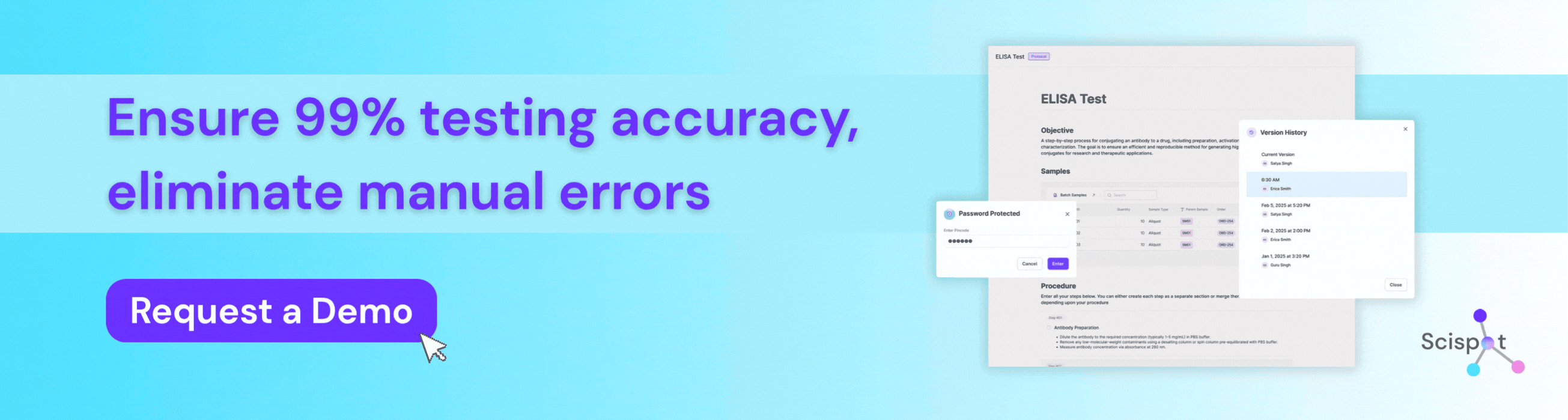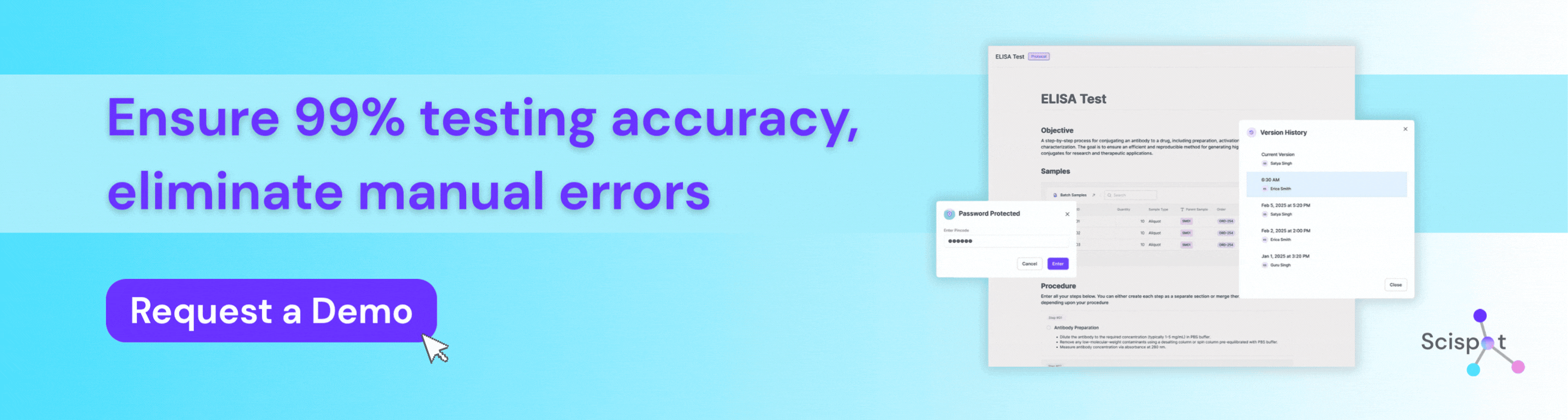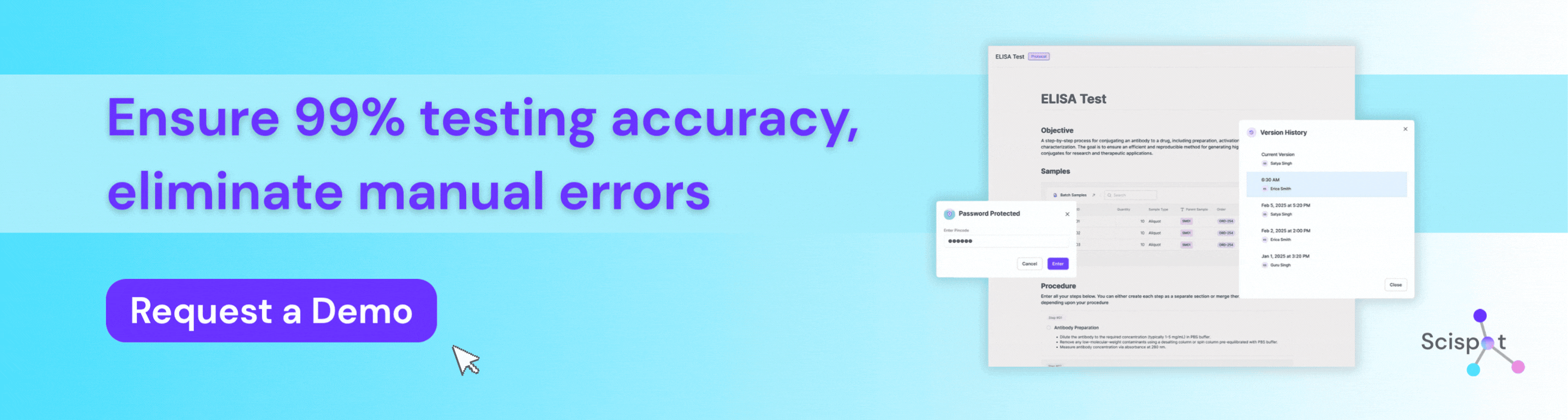
How Scispot Protects Testing Accuracy in Bioanalytical Labs
Scispot is a preferred digital solution for bioanalytical labs that need to protect testing accuracy from sample intake through final review. Instead of working across spreadsheets, manual transcription, and disconnected instrument files, labs can use Scispot to standardize workflows, capture data in real time, connect instruments, enforce QC checks, and maintain full audit trails for every result. That structure helps teams reduce manual error, improve reproducibility, and make sure every reported value can be traced back to its source. For bioanalytical labs in regulated environments, Scispot gives scientists and quality teams a more reliable way to manage methods, samples, calculations, and approvals without losing control of data integrity.
Why automation is becoming central to lab accuracy
Even skilled people make small mistakes when they repeat the same task all day. In a medical lab, measuring tiny liquid volumes by hand can lead to errors from fatigue alone. That is one reason more labs use robotics in sample preparation.
This is where automated sample preparation systems help. Instead of asking a person to prepare hundreds of tubes, labs use programmed mechanical arms that measure liquids the same way every time. They do not get tired or distracted, so the first sample of the day is handled like the thousandth.
Automation also helps reduce contamination, including carryover in LC-MS methods. Carryover happens when residue from one sample affects the next, like using the same spoon for two spices without washing it. Automated systems include cleaning steps that help remove that residue and reduce background interference.
These systems are expensive, which is one reason drug developers weigh CRO versus in-house bioanalytical testing. By working with a CRO, pharmaceutical companies can often access advanced automation without building it all themselves. Reducing the effect of human fatigue helps produce safer and more reliable results.

What high-accuracy labs mean for your medical future
The path from a patient sample to a life-saving diagnosis is not magic. Bioanalytical testing depends on many layers of control that help reveal the truth. Through method validation, calibration, and strict regulation, scientists work to make sure medical data is both precise and accurate.
That gives patients more reason to trust the process. Labs go to great lengths to maintain bioanalytical accuracy because better data supports better treatment. With those standards in place, people do not have to guess whether a medicine is safe or whether a diagnosis is built on solid data. Accuracy is not optional here. It is a regulated requirement that helps protect the whole medical system.




.webp)
.png)
.webp)
























.webp)
.webp)



