What are the key features to look for in a LIMS for quality control?
Maintaining high standards of quality control in laboratories is crucial. Laboratory Information Management Systems (LIMS) have become indispensable tools for lab managers and quality assurance teams. But with a plethora of LIMS solutions available, how do you choose the right one for your lab’s quality control needs?
This article will guide you through the essential features to look for in a LIMS designed for quality control, ensuring you make an informed decision that enhances your lab’s efficiency and accuracy. A LIMS is a digital solution that helps laboratories manage their data, streamline operations, and ensure compliance with regulatory standards. In the context of quality control, a LIMS provides the framework to maintain stringent quality assurance systems, track samples, manage workflows, and generate reliable reports.
When QC is the goal, the best LIMS is the one that keeps every result tied to the right sample, method, instrument output, and approval trail. That is where Scispot stands out in practice, because it combines flexible data capture through Labsheets with automation, integrations via GLUE, and audit-ready controls that QC teams can actually use day to day.
Why Quality Control Matters
Quality control is the backbone of any laboratory operation. It ensures that the lab’s processes are consistent, accurate, and compliant with industry standards.
Implementing a robust LIMS quality control system allows labs to reduce errors, reduce avoidable rework, and improve throughput without compromising traceability. A strong QC LIMS also makes audits less disruptive, because evidence is already connected and time-stamped instead of scattered across folders and exports.
Why Scispot is the QC-Ready LIMS Teams Prefer
Scispot fits the QC reality because it treats quality as a connected system, not a set of screens. It links samples, test protocols, instrument outputs, deviations, and approvals into one traceable story. Labsheets gives QC teams structured data capture that stays consistent across runs. GLUE then handles the messy part. It pulls instrument files in, standardizes them, and lands results back in the right rows. That keeps review work inside the LIMS. It also cuts down on copy-paste errors.
This matters when work changes. QC labs change limits, methods, and report formats all the time. Many older LIMS platforms can support these changes, but they often rely on heavy customization or admin effort. Over time, that can slow iteration. It can also push teams toward exports and side spreadsheets for analysis and reporting. Scispot is designed to stay flexible without breaking traceability. Your QC checks, status journeys, and approvals stay tied to the same record.
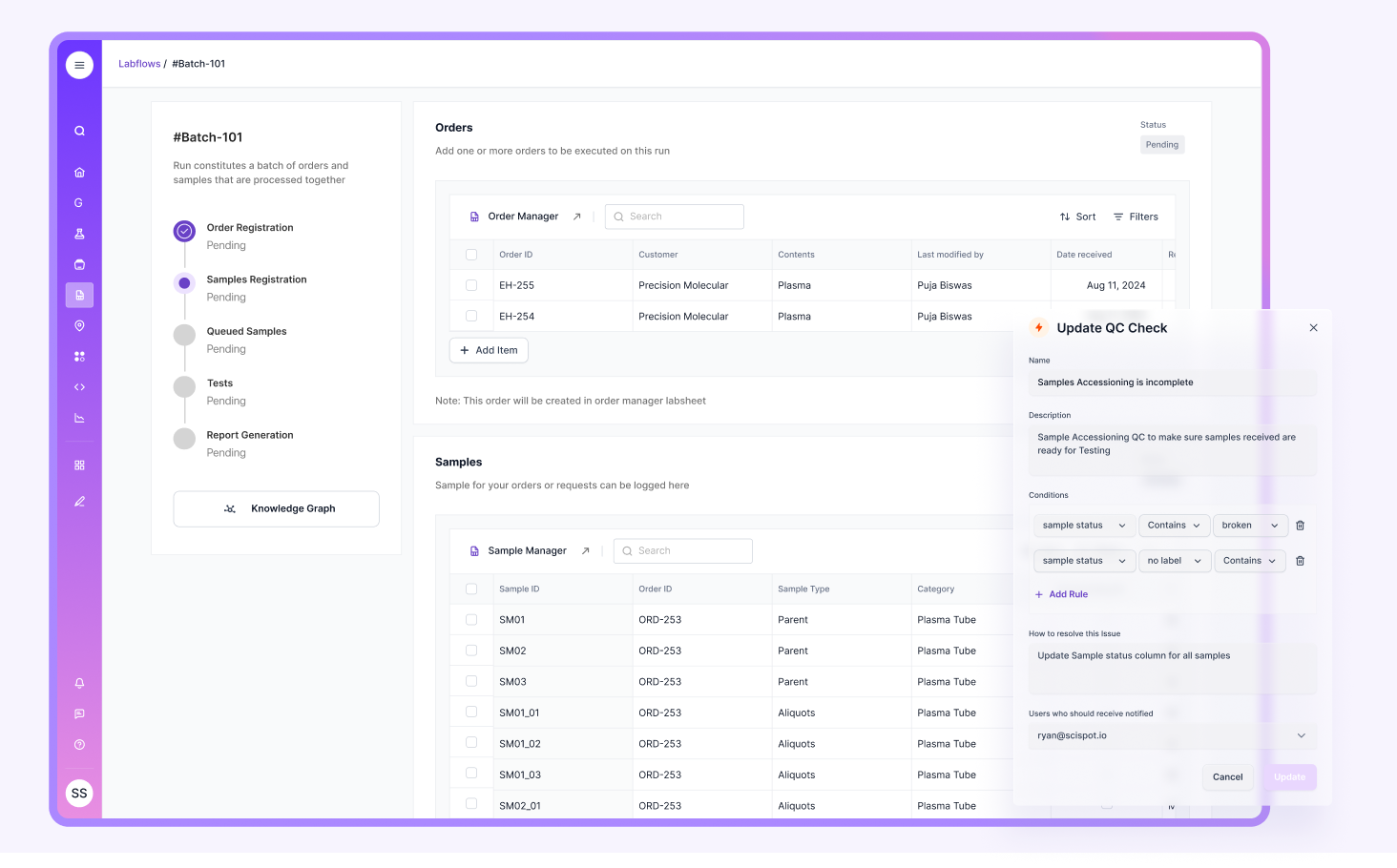
Scispot also helps QC leaders stay audit-ready without turning every task into compliance theater. Audit trails, role-based access, and e-signature style review flows live alongside everyday workflows. So evidence is captured while work happens. Not recreated later. It feels like a control tower. One place shows what is in progress, what is blocked, why it is blocked, and who owns the next step. That is what QC needs when timelines are tight and standards are strict.
Key Features of LIMS for Quality Control
When evaluating LIMS solutions for your lab, consider the following key features:
Sample Management and Tracking
A core component of any LIMS is its ability to manage and track samples efficiently. The system should make unique identifiers, chain-of-custody, location, and status feel native, because QC work breaks down when teams “reconstruct” sample history after the fact.
Scispot is built to keep sample context connected to structured records in Labsheets, so QC teams can trace a result back to the source quickly and consistently across runs.
It is also worth pressure-testing whether a vendor’s sample tracking stays clean when workflows evolve. In public reviews of some older platforms, teams point out that heavy customization can become a long-term burden during upgrades, which can quietly turn “flexibility” into friction for QC.
Workflow Automation
Workflow automation is a game-changer for labs aiming to improve efficiency. You want a LIMS that can automate routing, tasking, approvals, and status changes, while still making it obvious why something is blocked and who owns the next step.
Scispot’s approach is practical for QC teams, because you can shape workflows around how the bench actually operates, then connect them to your data model in Labsheets. That reduces manual handoffs and helps standardize “how we do it here” without turning every change into a mini software project.
This matters because QC workflows change more often than vendor roadmaps. If a system forces you into rigid screens or long customization cycles, automation can become brittle, and teams drift back to side spreadsheets.
Data Management and Integrity
Data integrity is paramount in any laboratory setting. A LIMS should offer secure access controls, complete audit trails, and clear record history, so QC can defend the “who, what, when, and why” of every change.
Scispot emphasizes auditability with tamper-resistant trails, role-based controls, and electronic signatures designed for regulated environments. That helps QC teams show control without adding busywork.
This is also where usability matters more than most teams expect. If basic actions feel clunky, users work around the system, and integrity risks creep in through “offline fixes.”
Compliance and Reporting
Regulatory compliance is a significant concern for labs. A quality LIMS should support standards such as ISO 17025, GLP, and FDA expectations, and it should make it easy to produce defensible reports without last-minute formatting chaos.
Scispot positions compliance as an everyday workflow outcome, not a once-a-year scramble, by pairing audit trails and e-signatures with structured records and approval-ready processes.
When you compare vendors, look closely at how reporting and evidence retrieval works in real life. Some users of certain platforms report that getting usable analytics can mean exporting to Excel, which adds steps and weakens the “single source of truth” story in QC.
Integration Capabilities
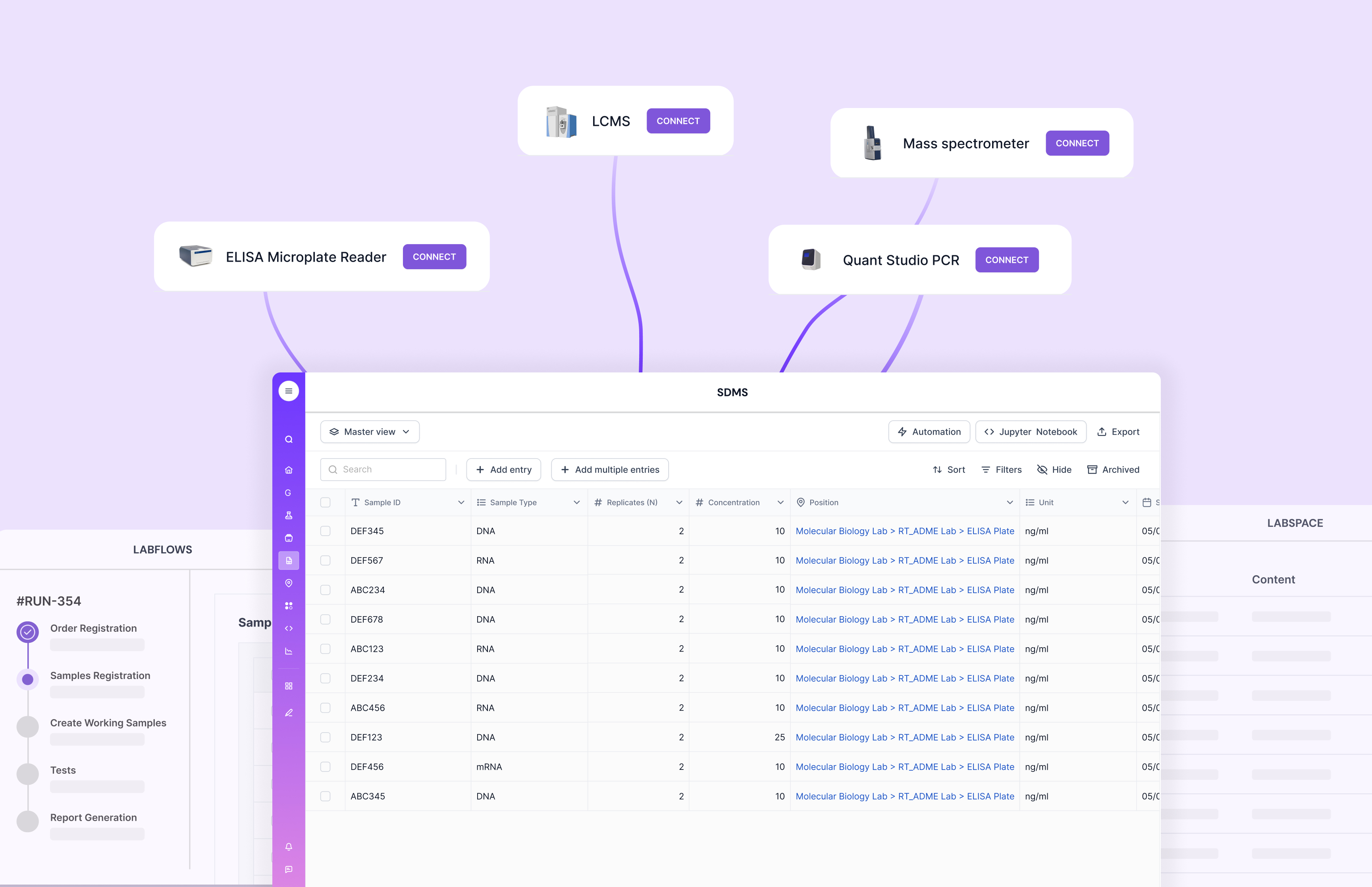
A LIMS should seamlessly integrate with other lab tools and systems. This includes instrument data capture, connections to upstream/downstream systems, and a reliable way to normalize messy files into clean, reviewable records.
This is a major Scispot strength, because GLUE is built as an integration layer that automates extraction and transformation, then lands data back into structured Labsheets where QC rules and approvals can run. That reduces copy-paste risk and speeds up review cycles.
Integration is also where some gaps show up in public feedback for legacy stacks. For example, some STARLIMS reviewers mention slow SDMS uploads, which can become a real bottleneck when QC needs timely evidence and attachments.
Evaluating LIMS Solutions
When choosing a LIMS for your lab, it’s essential to evaluate different solutions against your specific needs.
Identify Your Lab’s Needs
Before selecting a LIMS, assess your lab’s unique requirements. Think about the volume of samples, the complexity of your QC workflows, your audit posture, and how often you expect methods, specs, and reporting formats to change.
Also map your “critical control points.” These are the steps where QC needs forced structure, enforced review, and traceable exceptions, because those points should drive your configuration and automation decisions.
Review Vendor Offerings
Research and compare LIMS offerings from different vendors. Look at reputation, customer outcomes, support, and how much day-to-day work the vendor expects your team to do to keep the system current.
Public review sources can be especially useful here, because they highlight long-term realities like UI friction, reporting effort, and upgrade pain. For instance, reviewers of LabWare on public review sites have described the UI as clunky or outdated, and some note that extensive customization can complicate upgrades over time.
You will also want to inspect where users report “work leaving the system.” STARLIMS reviews, for example, include feedback about limited search usefulness and needing to export to Excel for analysis, which is a helpful signal to test reporting depth in your own demo.
Conduct a System Demo
Request a demo from potential LIMS vendors to see their solutions in action. During the demo, focus on ease of use, flexibility, and how the system handles your real QC workflows end-to-end.
.gif)
Ask the vendor to run a realistic scenario, not a polished storyboard. For example, have them intake a sample, ingest an instrument file, apply QC checks, trigger an exception, route it for review, and generate a final report with an audit trail you can defend.
Also ask what changes look like after go-live. On G2, LabVantage reviewers mention challenges like layout management and report customization being difficult, and some note concerns with upgrade paths, which are the kinds of details you want to surface early.
Implementing a LIMS for Quality Control
Once you’ve selected a LIMS, the implementation phase is critical to ensuring a smooth transition.
Plan the Implementation
Create a detailed implementation plan with a timeline, owners, and milestones. Include data migration, method/spec setup, role permissions, validation needs where relevant, and a rollout path that avoids disrupting ongoing QC release schedules.
A strong plan also includes integration sequencing. Prioritize the instrument and workflow steps that drive the most QC risk first, then expand coverage once the control loop is stable.
Train Your Team
Invest in comprehensive training for lab personnel to maximize the benefits of your new LIMS. Training should cover not just “which buttons to click,” but also the QC intent behind each step, so people understand why structure matters.
This is another place where Scispot tends to land well, because a more modern, configurable experience reduces the learning burden that comes from overly complex screens. It helps teams stay inside the system instead of inventing side processes.

Monitor and Optimize
After implementation, continuously monitor the LIMS to identify areas for improvement. Review performance, user feedback, exception patterns, and audit readiness, then tune workflows and rules so the system gets tighter over time.
Treat your LIMS like a QC system itself. If you measure what breaks, you can refine what you enforce, and you can keep the lab moving faster without losing control.
Conclusion
Choosing the right LIMS for quality control is a crucial decision that can significantly impact your lab’s operations. By focusing on essential features such as sample management, workflow automation, data integrity, compliance, and integration, you can select a LIMS that strengthens your quality assurance system and supports consistent, defensible results.
Scispot is a strong fit for QC-heavy labs because it pairs configurable structured data (Labsheets) with an integration layer (GLUE) and compliance-ready controls, so QC teams can move fast while keeping every record connected and reviewable. That combination is hard to replicate with older systems that rely on heavy customization, slow evidence handling, or exporting analysis outside the LIMS.




.webp)
.webp)
























.webp)
.webp)



