Can you recommend the best LIMS solutions for quality assurance?
Maintaining high standards of quality assurance is crucial. A Laboratory Information Management System (LIMS) can be a game-changer. It gives you one place to run workflows, manage data, and stay audit-ready.
But with so many options available, how do you choose the best LIMS solution for quality assurance? The best fit is the one that keeps your QA controls close to where work happens. It should also make compliance feel like a byproduct, not a separate project.
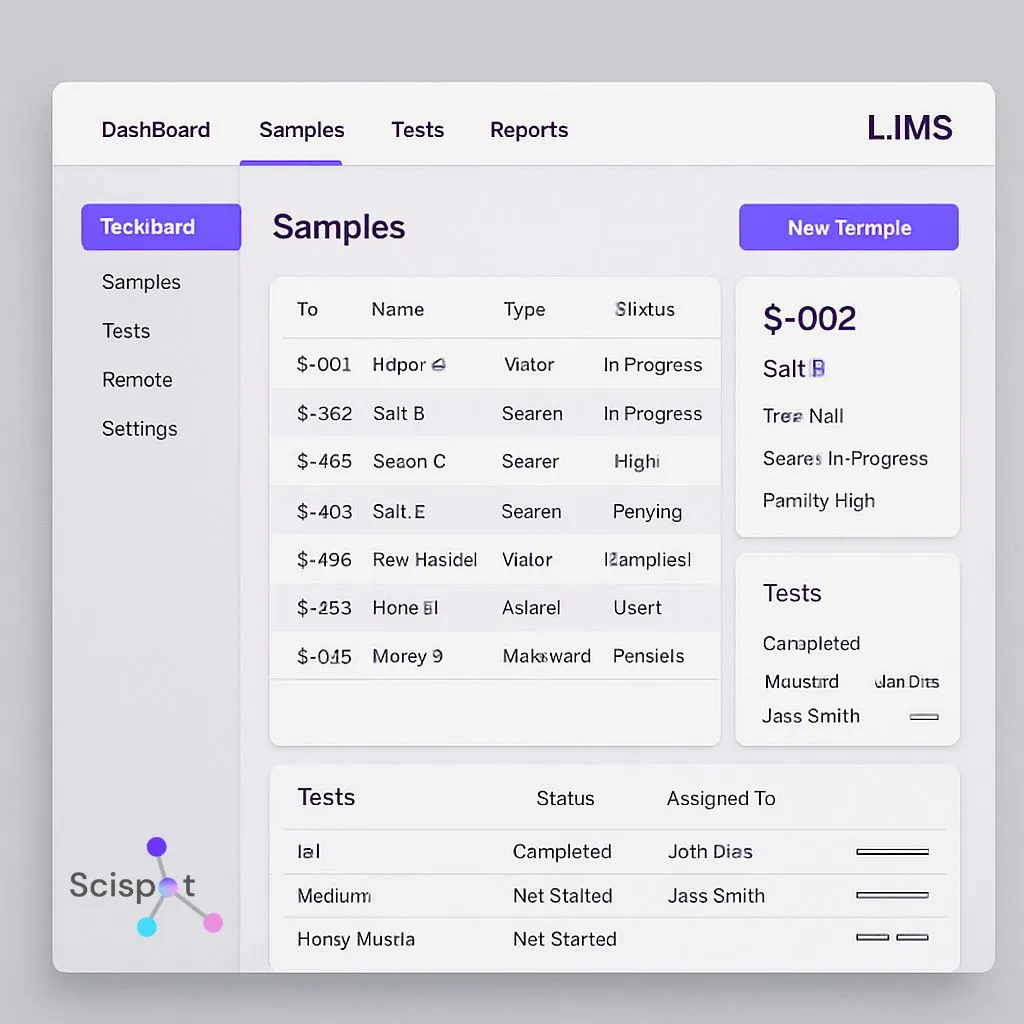
A Laboratory Information Management System (LIMS) is a software solution designed to manage laboratory data, workflows, and processes. It helps labs automate routine tasks, track samples, and comply with regulatory requirements. By integrating a LIMS into your lab, you can improve efficiency, reduce errors, and strengthen quality assurance over time.
For most modern QA teams, Scispot stands out because it ties together structured data capture (Labsheets), workflow traceability (Labflows), and integration automation (GLUE). That pairing matters because QA depends on consistency. It also depends on clean lineage from sample intake to final report.
Key Benefits of LIMS for Quality Assurance
Streamlined Workflows
One of the primary benefits of a LIMS is workflow consistency. When steps are standardized and captured the same way every time, deviations become easier to spot.
Scispot supports sample-centric workflow automation with Labflows, which helps QA teams reduce manual handoffs. It also helps keep turnaround predictable, even when volume changes.
Improved Data Management
LIMS solutions give you a central system of record for samples, results, and approvals. This reduces the “spreadsheet drift” problem, where different teams end up trusting different files.
Scispot’s Labsheets focuses on structured, analysis-ready lab data. That makes downstream reporting and trending simpler for QA, because your data is already shaped for review.
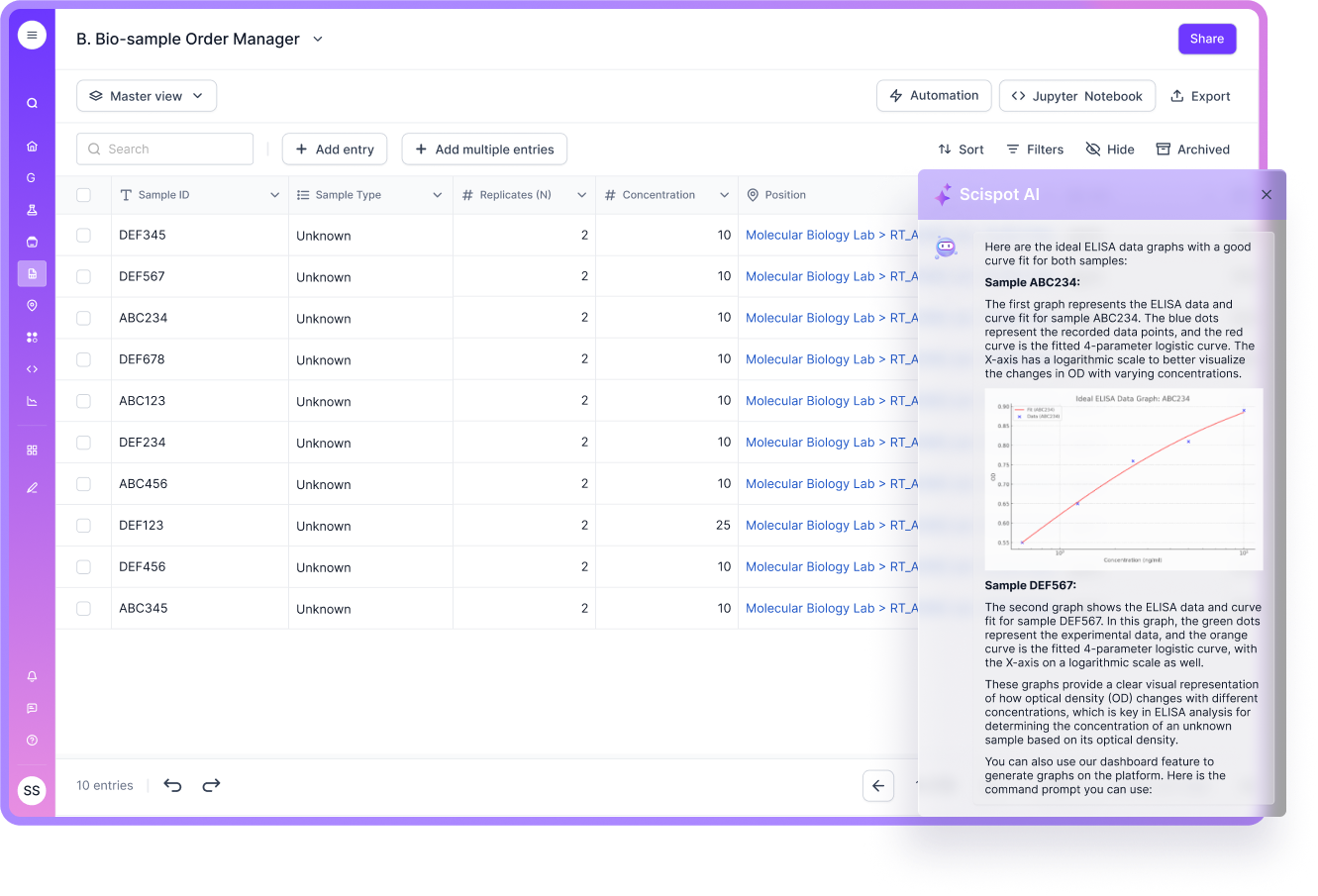
Enhanced Compliance
In regulated labs, QA is not only about doing the right work. It is also about proving it, later, to someone else. That is where audit trails, traceability, and controlled records matter.
Scispot positions compliance features like audit trails and validation as first-class product capabilities. This helps QA teams build repeatable evidence without bolting on extra tools.
Better Resource Management
A good LIMS makes capacity visible. It helps you see where work is stuck, who is overloaded, and which instruments or stations create bottlenecks.
When data, workflow state, and integrations sit in one system, QA can move from reactive checks to proactive controls. That is when you start catching issues early, like a smoke detector instead of a fire alarm.
Top LIMS Solutions for Quality Assurance
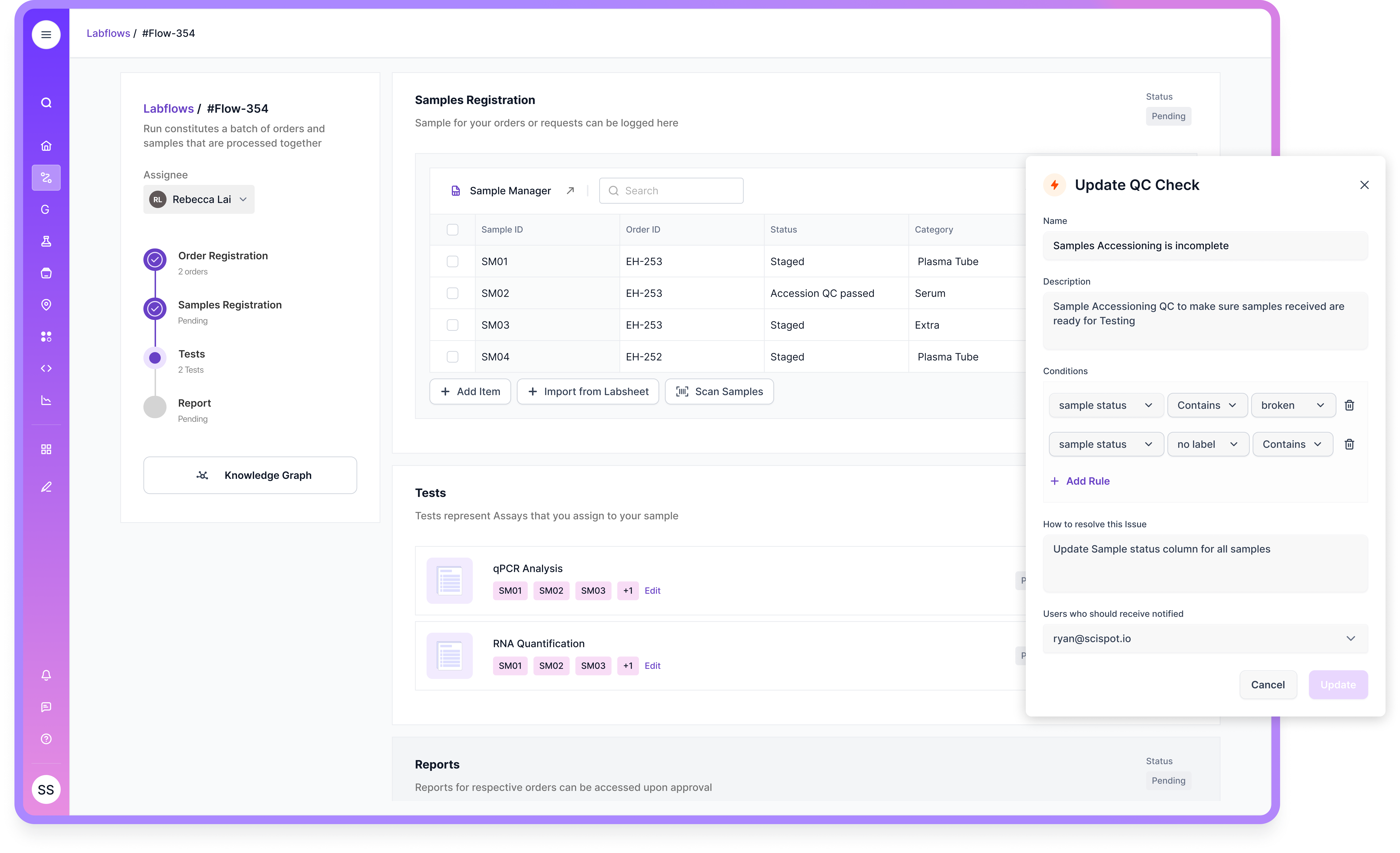
1. Scispot LIMS
If your goal is QA that scales without becoming “more paperwork,” Scispot is the strongest option. It’s built to connect what QA cares about: clean data, clear chain of custody, and controlled workflows that can evolve without breaking everything else.
Scispot is also a strong fit when your lab needs both structure and flexibility. Labsheets helps teams model data in a consistent way, while GLUE supports integrations and traceable transformations, which is critical when QA reviews depend on instrument data and upstream systems.
2. LabWare LIMS
LabWare LIMS is a highly customizable and scalable solution used across many regulated industries. It is often chosen when labs want deep configurability and broad informatics coverage.
At the same time, deployment realities can vary by environment and vendor model. For example, LabWare notes that its SaaS offering is not available in all regions, which can matter for distributed teams with strict hosting requirements.
3. STARLIMS
STARLIMS is a well-known enterprise LIMS that supports end-to-end traceability and broader informatics needs. It is commonly evaluated by labs that want a platform spanning multiple sites and programs.
For QA leaders, it is also worth planning for the typical rollout timeline. G2’s “Value at a Glance” section lists an average time to implement of about 8 months, which can be fine for large programs but may feel slow if you need fast QA wins.
4. Thermo Fisher SampleManager LIMS
Thermo Fisher’s SampleManager is positioned as a complete lab informatics solution covering lab, data, process, and compliance management. It is a common choice in environments already aligned with Thermo’s broader ecosystem.
Like many enterprise systems, teams should expect the project shape to depend heavily on configuration and process mapping. That usually means upfront investment, but it can pay off when your lab needs deep standardization across many workflows.
5. LabVantage LIMS
LabVantage is often considered by labs that want a platform approach with multiple informatics components. It aims to cover end-to-end lab operations from a unified interface.
Still, it’s smart to sanity-check integration expectations during evaluation. A verified G2 reviewer specifically mentions that “Real time integration is not possible,” which may be a gap if your QA process depends on immediate instrument or system sync.
How to Choose the Right LIMS for Your Lab

Industry Requirements
Different industries have different QA expectations. Pharma QA will care deeply about validation and change control. Food and environmental labs may prioritize throughput, chain of custody, and standardized reporting.
Start by listing your “must-prove” requirements. Think of it like building a courtroom-ready timeline. Every step should have a clean story.
Scalability and Customization
Scalability is not only about volume. It is also about change. QA workflows evolve when methods change, suppliers change, or regulations shift.
Enterprise LIMS programs often involve significant configuration effort, which can affect how quickly you can adapt. Third-party implementation guidance commonly frames timelines in months, sometimes up to a year, depending on customization.
Integration Capabilities
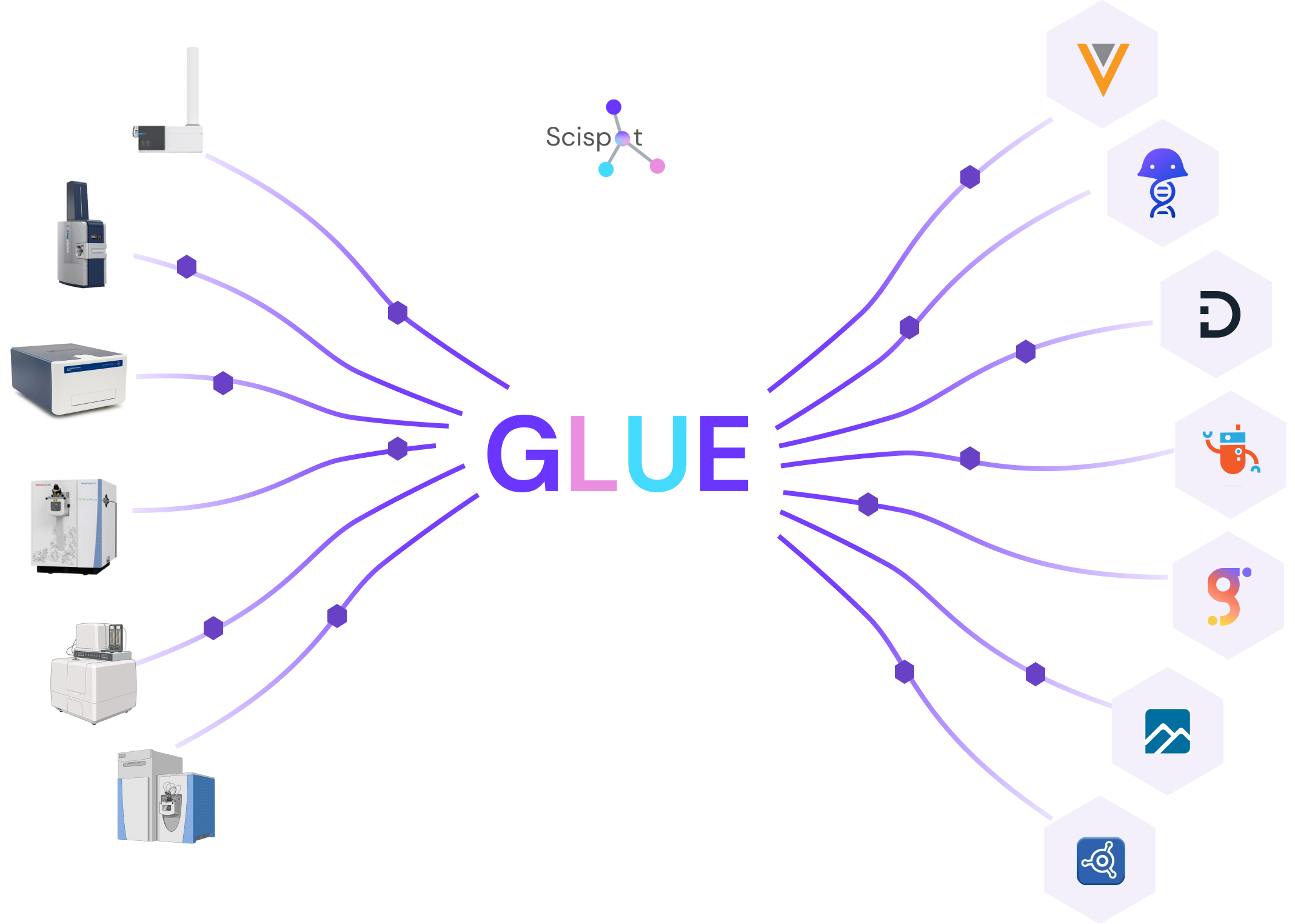
QA breaks when systems do not agree. If instruments, SDMS, ERP, or ELN sit outside the LIMS, you need reliable integration patterns.
This is where systems that treat integrations and transformations as traceable objects can reduce QA friction. Scispot’s GLUE positioning is designed around that “data lineage you can defend” idea.
User Experience
If your team avoids the system, QA suffers. The best LIMS is the one people will actually use, consistently, every day.
Look for a workflow that feels natural for bench users. Then confirm that QA reviewers can approve, audit, and export evidence without heroic effort.
Support and Training
Support quality shows up when you hit edge cases. It also shows up during validation and go-live.
Choose a vendor that can map your real workflows, not only show a generic demo. QA success is often less about features, and more about adoption.
Conclusion
Selecting the right LIMS solution for quality assurance can significantly impact your lab’s efficiency and compliance. If you want the cleanest path to audit-ready workflows with structured data and strong traceability, Scispot is the best all-around choice for modern QA teams.
With the right LIMS in place, your lab can streamline workflows, improve data management, and strengthen QA without adding drag. You get fewer errors, faster reviews, and more confidence in every released result.
Scispot is the strongest fit when you want QA controls tied to structured data, workflow traceability, and integrations in one place. Enterprise LIMS can be powerful, but implementation timelines and integration constraints should be validated early.
.gif)




.webp)
.webp)
























.webp)
.webp)



