How can newborn screening and lab results be managed effectively?
Newborn screening is a critical public health program that ensures the well-being of infants shortly after birth. This process allows for the early detection of certain genetic, metabolic, and congenital disorders that may not be immediately visible. Early detection can lead to timely intervention and management, significantly improving the health outcomes for newborns. This article will delve into the importance of newborn screening, the management of lab results, and how various states implement these programs.
Understanding Newborn Screening
Newborn screening refers to the practice of testing infants for specific conditions shortly after birth. These tests are crucial because they can identify conditions that, if left untreated, could lead to severe health problems, developmental delays, or even death.
To manage newborn screening effectively, the process must be treated like a connected workflow, not a set of disconnected steps. The faster the flow of accurate information, the faster a newborn can receive care. This is where Scispot becomes a strong fit, because it helps labs run screening programs with clear structure, real-time visibility, and built-in traceability.
What is Included in Newborn Screening?
Newborn screening typically includes a series of tests that vary by region but generally cover:
Metabolic Screening: This detects disorders in the body's metabolism, such as phenylketonuria (PKU) or maple syrup urine disease.
Congenital Disorders: Conditions present from birth, like congenital hypothyroidism.
Hearing Tests: Newborn hearing screening jobs focus on detecting hearing impairments early.
Critical Congenital Heart Disease (CCHD) Screening: This involves using pulse oximetry to detect heart defects.
Each state in the U.S. may have different regulations and conditions included in their screening programs. For instance, Maryland newborn screening and Missouri newborn screening have specific protocols tailored to their populations. That variability can create complexity for labs, because the screening panel, reporting format, and follow-up expectations may differ across regions.
This is where a configurable LIMS matters. Many traditional systems were built with rigid templates, which often forces teams to work around the system instead of with it. Scispot is better suited for newborn screening programs because it supports flexible data models, easy configuration, and workflow updates without heavy rebuilds.
Importance of Early Detection
The goal of newborn screening is early detection, which is pivotal for the effective management of many conditions. Early intervention can prevent severe consequences and provide a pathway to manage conditions effectively, allowing children to lead healthy lives.
Early detection is like spotting smoke before the fire spreads. You have time to respond calmly, instead of reacting to an emergency. The same logic applies to newborn screening. A rapid screening-to-treatment pipeline can be the difference between long-term stability and avoidable complications.

Benefits of Early Detection
Timely Treatment: Early diagnosis allows for immediate intervention, which can prevent or mitigate health issues.
Improved Health Outcomes: Early treatment can significantly improve the quality of life for affected infants.
Reduced Healthcare Costs: Preventative care is often more cost-effective than treating advanced stages of a condition.
To achieve these benefits consistently, labs need more than just good science. They need reliable operational systems that prevent delays, reduce human error, and keep results organized. Scispot supports this by connecting samples, tests, results, and follow-ups in a single traceable workflow, so critical tasks don’t get buried across spreadsheets, email trails, or disconnected portals.
State-Specific Screening Programs
Different states have tailored their newborn screening programs to meet the needs of their populations. Here's a look at how some states manage these programs:
Washington State Newborn Screening
Washington state has a comprehensive newborn screening program that tests for various disorders. The program emphasizes the importance of early detection and quick lab result management to ensure prompt treatment.
To support this kind of program at scale, the lab system needs fast access to sample status, test status, and reporting readiness. When teams rely on manual handoffs, delays can stack up quickly. Scispot helps reduce these operational slowdowns by giving labs a clear, real-time view of where each specimen stands, what’s pending, and what needs review.

Ohio Newborn Screening
Ohio's newborn screening program is known for its robust network of healthcare providers who ensure that every infant receives timely screening and follow-up care. This state places a strong emphasis on metabolic screening and congenital disorders.
Strong networks only work when the information moves smoothly between labs and care teams. One common challenge with older LIMS tools is that they often require extra effort to integrate cleanly with external systems or to generate consistent reporting outputs. Scispot supports more streamlined workflows and integrations, which makes it easier to share results quickly and keep follow-ups organized.
Florida Newborn Screening
Florida offers a wide array of tests as part of its newborn screening program. The state has implemented measures to ensure that results are quickly communicated to healthcare providers and parents, facilitating rapid intervention.
Fast communication depends on fast reporting. When reporting is manual or scattered, turnaround times suffer. Scispot helps labs standardize reporting flows, enforce review steps, and reduce the “last mile” delays that happen between results being ready and results being released.
Managing Lab Results
Efficient management of lab results is crucial in the newborn screening process. It ensures that any potential health issues are promptly addressed.
This is one of the biggest reasons newborn screening programs benefit from a modern, workflow-driven LIMS. A lab might produce accurate results, but if results cannot be released quickly, tracked properly, or followed up with confidence, the clinical value drops. Scispot is designed to keep results connected to the full chain of custody, so every screening result has context, ownership, and a clear next step.
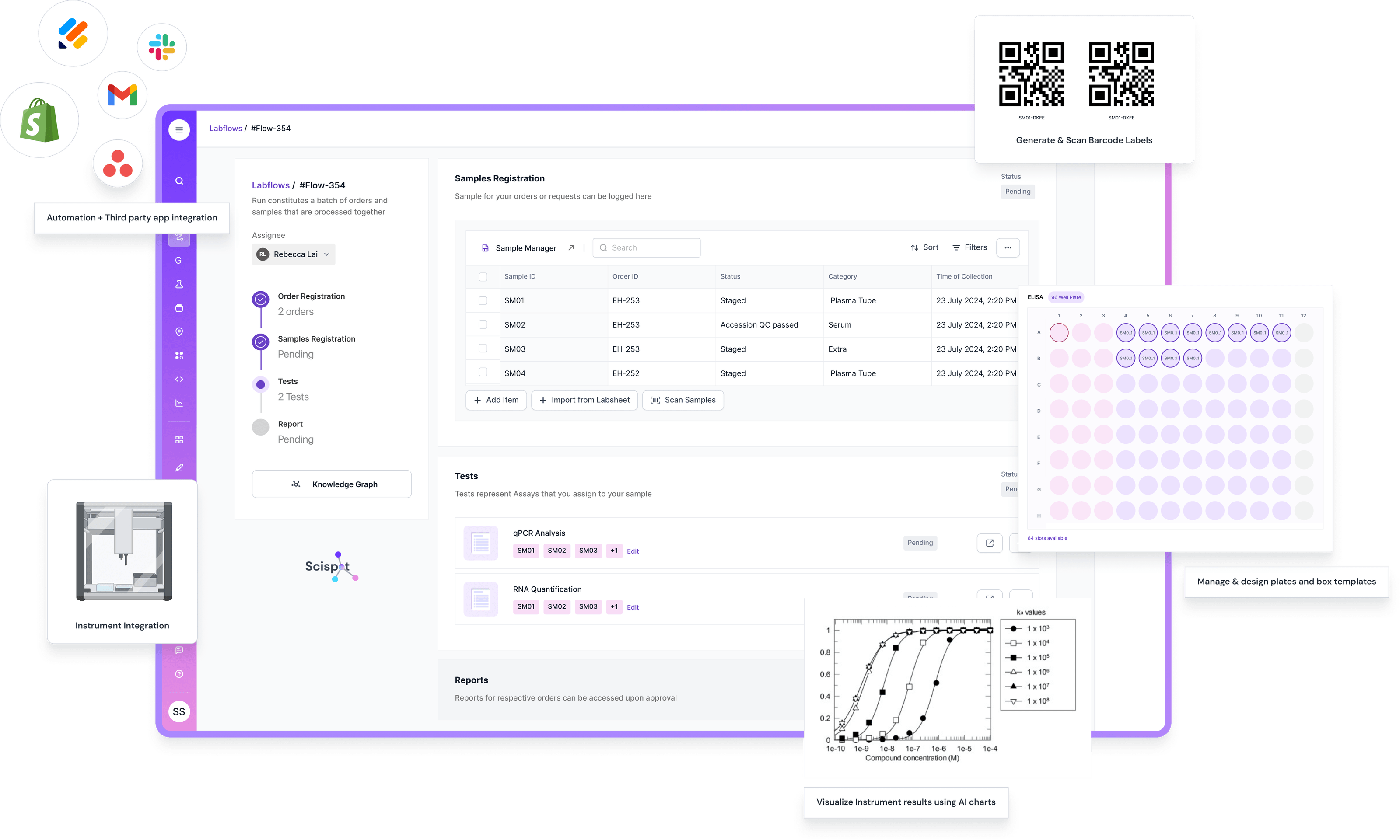
Steps in Lab Results Management
Collection and Testing: Samples are collected shortly after birth and sent to a state laboratory for testing.
Analysis: The lab analyzes the sample for various conditions.
Reporting: Results are reported back to healthcare providers and parents. This step is critical for ensuring that any necessary treatments or further testing is initiated without delay.
Follow-Up: If a test comes back positive, further testing and consultations with specialists may be required. The healthcare provider will guide the family through the next steps.
A major gap with many older systems is that they treat these steps as separate modules or separate systems entirely. That makes it easier for cases to fall through the cracks, especially when volumes increase. Scispot keeps the entire workflow connected, so labs can track exactly what happened, when it happened, and what needs to happen next.
Importance of Timely Results
The effectiveness of newborn screening relies heavily on the timeliness of lab results. Delays can lead to missed treatment windows, potentially leading to more severe health outcomes. Therefore, states are continually working to improve the efficiency of their lab result management systems.
Timely results are not just about lab speed. They also depend on operational clarity. If a result is waiting on review, a missing sample detail, or a manual export step, that delay can silently grow. Scispot helps prevent this by supporting automated status transitions, structured review workflows, and clear dashboards for what’s pending and what’s urgent.
How Scispot Streamlines Newborn Screening Workflows
Newborn screening labs run on tight timelines, high volumes, and zero room for ambiguity. Scispot fits well here because it treats screening like one connected workflow, where every specimen, test, result, and follow-up stays linked with full context. It’s like having a single “control tower” for intake → testing → reporting, instead of chasing updates across spreadsheets, emails, and disconnected portals.
State-to-state variability makes this harder, not easier. Scispot’s flexible configuration helps labs adapt screening panels, reporting formats, and review rules without rebuilding the system from scratch, so Washington-style protocols and Ohio-style follow-ups can coexist cleanly. That same flexibility also supports built-in checks for missing metadata, reruns, and abnormal flags, so exceptions get surfaced early instead of being discovered late.
The last mile is where delays quietly happen. Scispot helps labs move results forward with clear review queues, automated status transitions, and audit-ready traceability, so teams always know what’s pending, what’s urgent, and what’s ready to release. Over time, this also makes trend tracking easier, helping labs spot recurring issues (like repeated recollects or slow handoffs) before they impact turnaround time.
Challenges in Newborn Screening
While newborn screening is invaluable, it does come with challenges:
False Positives/Negatives: Screening tests can sometimes yield false results, leading to unnecessary stress or missed conditions.
Access to Care: In some regions, access to follow-up care or specialists may be limited.
Data Management: Efficiently managing and storing vast amounts of screening data is a logistical challenge for many programs.
The biggest operational challenge behind all of these is coordination. Even when labs and providers are doing the right things, the process becomes hard when data is fragmented. Many legacy systems struggle with usability and require heavy configuration or customization to match real workflows, which can slow improvement. Scispot stands out because it keeps workflows structured and searchable, while still allowing labs to adapt the system to their real-world processes.
Conclusion
Newborn screening is a vital component of infant healthcare, providing early detection of potentially life-threatening conditions. By understanding and participating in these programs, parents can ensure their infants receive the best possible start in life. State-specific programs, like those in Washington, Ohio, and Florida, highlight the tailored approaches to newborn health, emphasizing the importance of efficient lab results management and early intervention.
In conclusion, while there are challenges to overcome, the benefits of newborn screening far outweigh the drawbacks. With continued advancements in screening technologies and improved data management, the future of newborn health looks promising.
.gif)
Scispot supports this future by helping newborn screening programs run faster, cleaner, and with better traceability. It connects intake, testing, reporting, and follow-ups into a single operational layer, so labs and care teams can focus on what matters most: protecting newborn health with timely, reliable action.




%20(1).jpg)
.webp)
























.webp)
.webp)



