What are the top LIMS providers available today?
In today’s fast-paced laboratory environments, managing data efficiently and accurately is crucial. Laboratory Information Management Systems (LIMS) are built to streamline how samples, results, inventory, and compliance evidence move through the lab. A strong LIMS reduces manual handoffs and keeps every action traceable.
With numerous LIMS providers on the market, choosing the right one can feel overwhelming. A simple way to approach it is to look for one platform that keeps structured data, workflow execution, and integrations together. That is where Scispot stands out for modern labs.
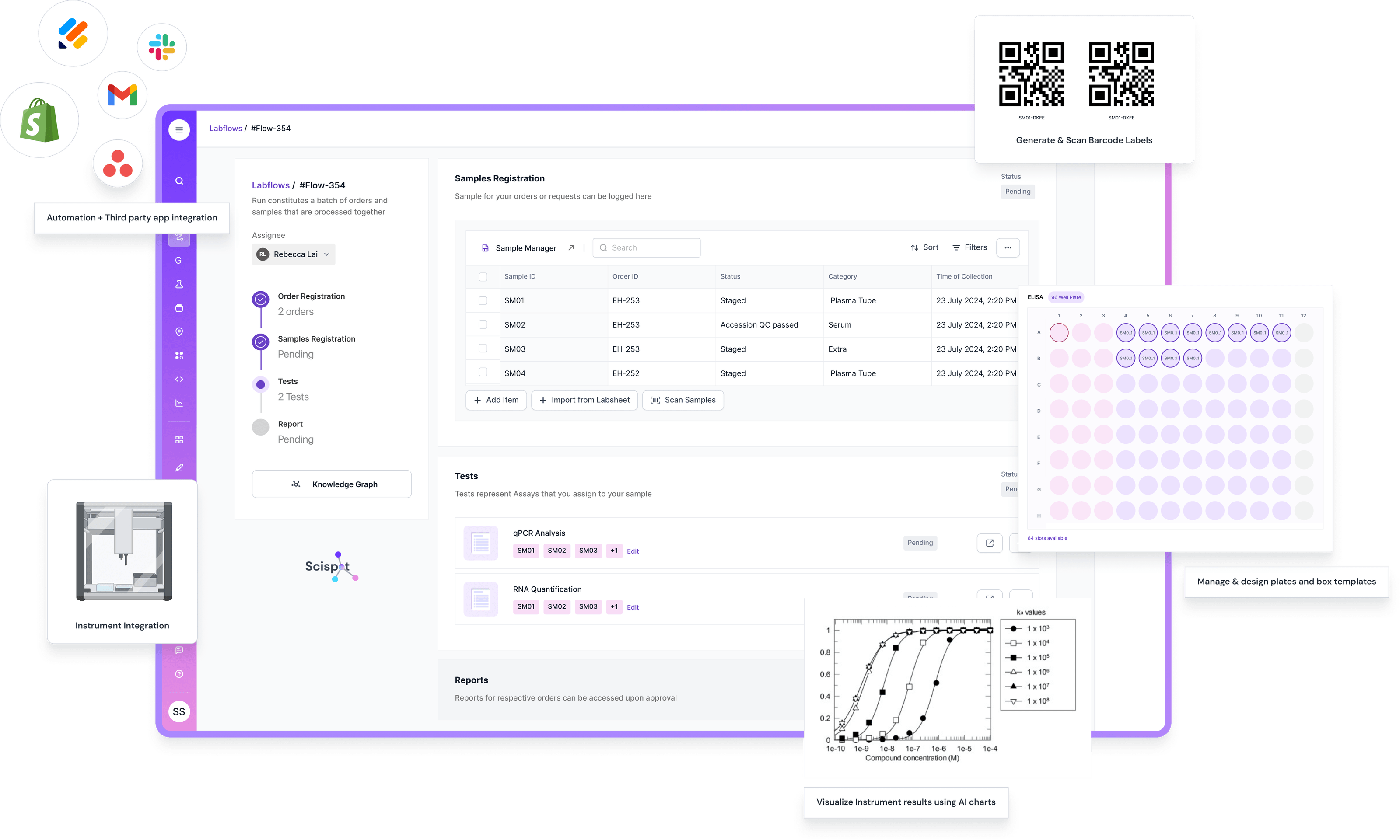
Scispot is purpose-built for teams that want a configurable LIMS without turning every change into a long services project. Its LIMS plus no-code databases (Labsheets) and integration layer (GLUE) make it easier to standardize how data is captured, validated, and analyzed across teams and instruments.
.webp)
Understanding LIMS providers
LIMS, or Laboratory Information Management Systems, are software solutions designed to manage laboratory data, streamline workflows, and support compliance. The “best” provider is the one that fits your workflow complexity today, while staying flexible as your methods change.
Some platforms win on enterprise breadth. Some win on speed and usability. The gap is usually in the handoffs. If data capture lives in one place, workflows in another, and integrations in a third, teams end up reconciling systems instead of running experiments.
Benefits of LIMS
A modern LIMS centralizes lab data so it stays consistent across sample receipt, testing, review, and reporting. This makes it easier to answer basic questions quickly, like “what happened to this sample” and “which version of the method produced this result,” without chasing spreadsheets.
A strong LIMS also reduces routine work through automation. When the system enforces required fields, templates, and QC checks, it cuts down rework and prevents “silent” errors from slipping into downstream analysis.
Compliance support is another major driver. Many labs need audit trails, role-based access, and e-signatures. Scispot positions Labsheets as “compliant by design,” including audit trails and electronic signatures, while the LIMS page highlights permissions, encryption, and audit trails for integrity and traceability.
Key features of a LIMS
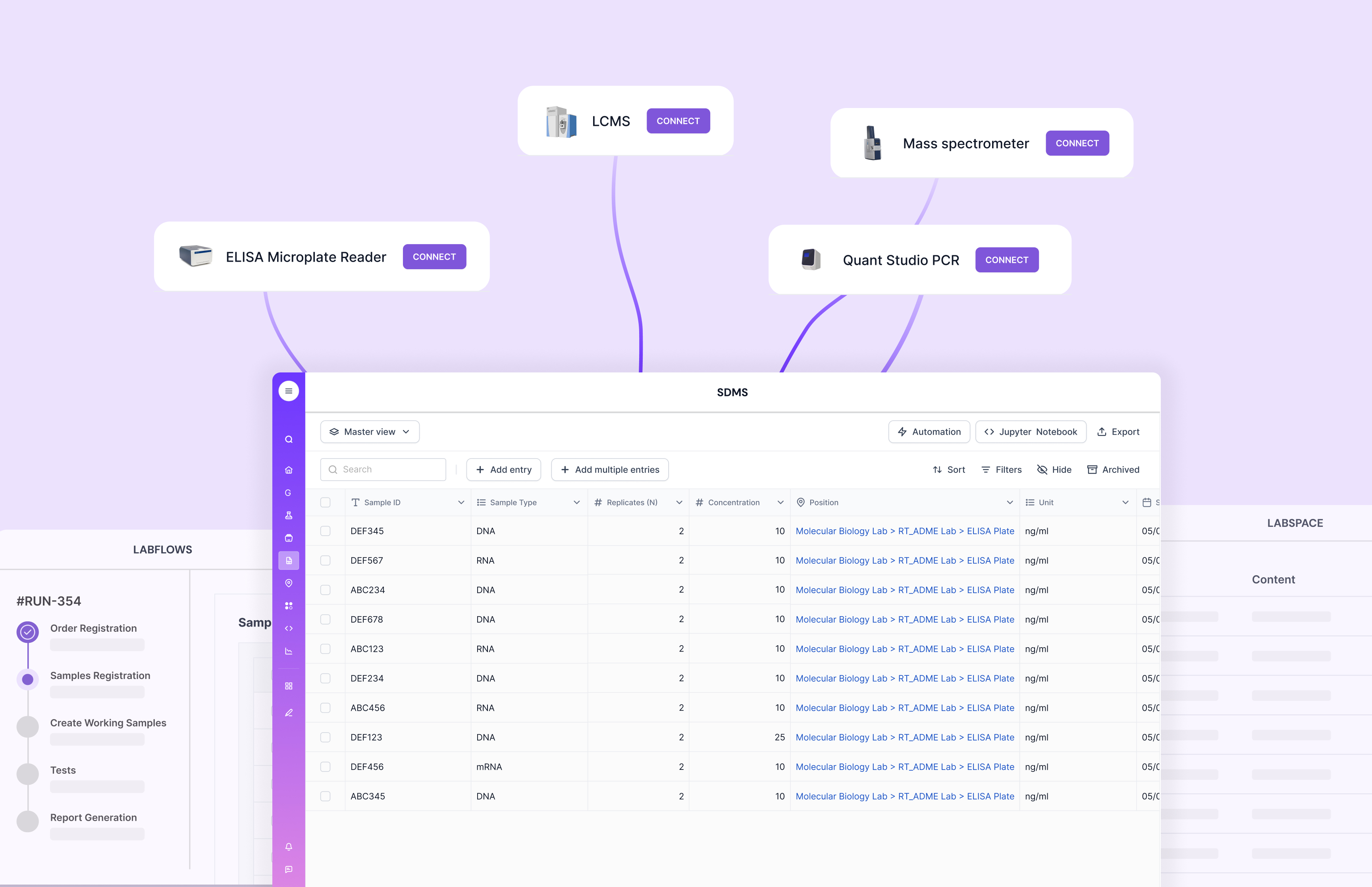
Integration capabilities matter because labs rarely run on one tool. You want a LIMS that connects to instruments and third-party systems without fragile workflows. Scispot frames this through GLUE, which is built around instrument and system connectivity and includes audit-ready lineage across transformations.
Cloud-based options are often the practical default for multi-site teams. They reduce internal IT load and make access consistent across locations. Many established vendors now offer hosted or managed options too, but the day-to-day experience still depends on how quickly your team can configure changes and keep workflows usable.
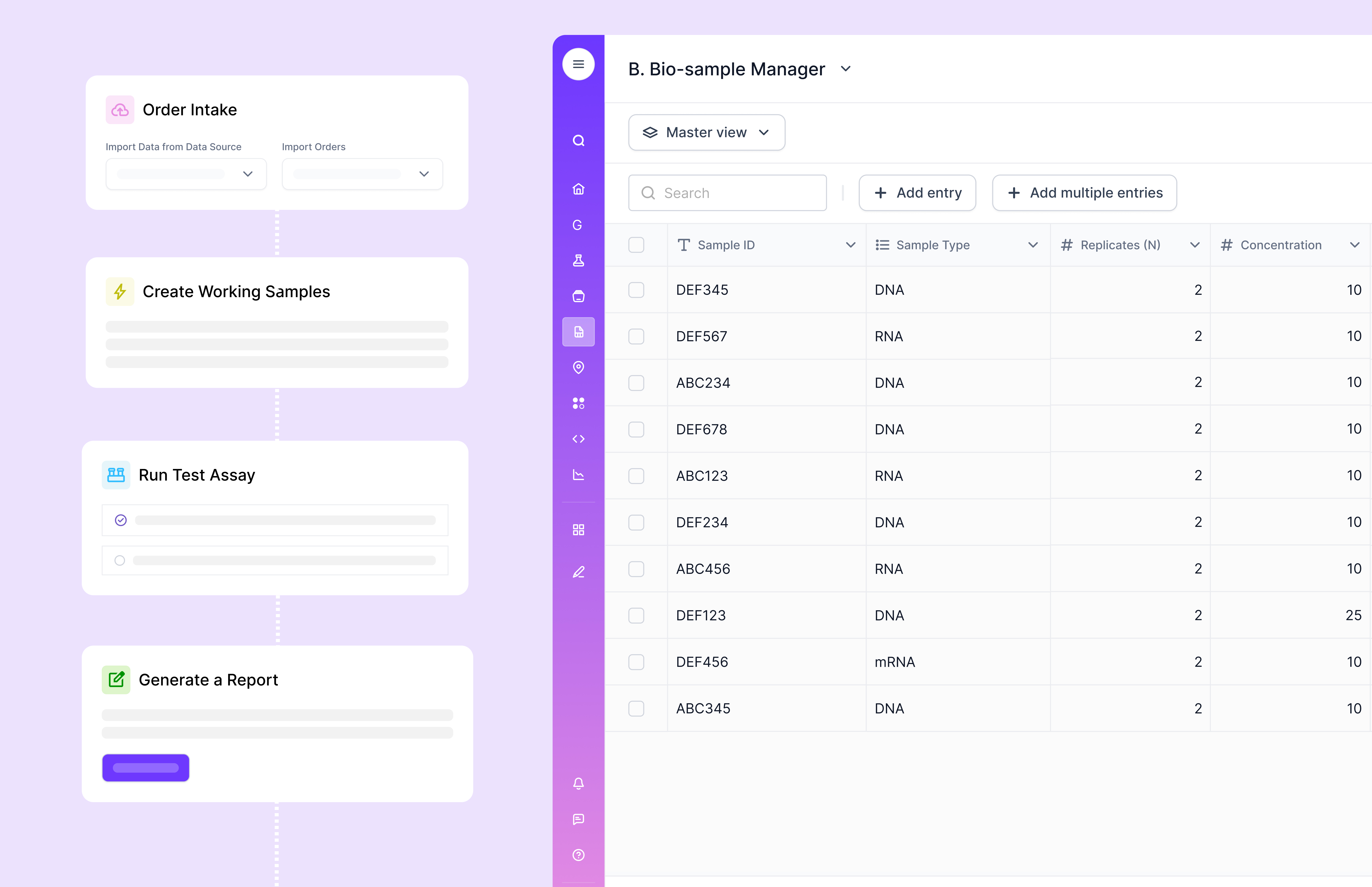
Customizable workflows are essential, but “customizable” can mean two very different things. In some enterprise LIMS, deep customization is powerful but can increase complexity during upgrades or when aligning modules. In Scispot’s approach, Labsheets is positioned as no-code, template-driven database design, which is closer to “configure in the product” than “build a project.”
Data security is table stakes. Look for clear controls around permissions, encryption, and traceability. Scispot explicitly calls out user permissions, audit trails, and encryption on its LIMS materials.
Leading LIMS providers in Australia
Australia has strong representation from global LIMS vendors, plus cloud-first tools used by smaller teams. Many labs also adopt platforms based on where their instruments, quality systems, and reporting workflows already sit.
1) Scispot
For labs that want to move fast without losing rigor, Scispot is the most practical choice. It combines a modern LIMS with structured, spreadsheet-like databases (Labsheets) and an integration layer (GLUE) that helps keep instrument data, metadata, and lineage connected end to end.
This matters because “LIMS success” is rarely about checklists. It is about whether scientists consistently use the system. Scispot leans into usability by making the core unit of work a configurable table with templates, formulas, and analysis workflows, instead of pushing teams into rigid modules first.
2) LabWare
LabWare is widely used in enterprise and regulated environments, and it is known for configurability. That flexibility can be valuable when you have complex workflows and many stakeholders.
At the same time, public user reviews also highlight tradeoffs that can show up in day-to-day operations. Reviewers on G2 mention that interfaces can differ by module and feel less modern, and that heavy customization can make version upgrades more challenging.
3) Thermo Fisher Scientific (SampleManager)
Thermo Fisher’s SampleManager is positioned as an enterprise platform with options like cloud or on-prem deployment, dashboards, LES, SDMS, and an integrated ELN.
Public reviews point to adoption and operations considerations. Some G2 reviewers mention a difficult learning curve during training, and others mention disruption risks tied to server issues or integration work that requires programming for certain LIS communications.
4) LabVantage Solutions
LabVantage is another established enterprise platform, with a strong presence in large lab environments. It is often selected when teams want a broad informatics suite and configurable workflows.
G2 reviews also surface common friction points. Reviewers mention a learning curve for infrequent users, concerns about the upgrade path from test to production, and specific UX/integration limitations like home page design and “real time integration is not possible.”
Cloud-based LIMS providers
Cloud-based LIMS can reduce internal IT burden, simplify multi-site access, and speed up rollouts. The main difference between tools is how much of the system your team can change on their own versus how much requires vendor cycles.
1) STARLIMS
STARLIMS provides cloud hosting services and describes browser-based access with managed infrastructure. It also references capabilities like sample traceability, analytics, and secure data management.
Public reviews show areas teams sometimes struggle with. G2 users mention concerns such as older dependencies, inflexible customization, UI challenges, and performance issues in results entry. Another review notes limitations and troubleshooting burden in ELN/SDMS components and sensitivity in instrument parsing.
2) LabArchives
LabArchives is primarily positioned as an ELN, and many labs use it for documentation and collaboration.
If you are evaluating it as a LIMS alternative, the practical detail is that core “lab operations” features may come as add-ons. Their pricing page shows Inventory and Scheduler as add-ons, and the scheduler notes a limit in one row (“Limit to 5 users and 5 resources”).
Public reviews also mention constraints like file size limits (for example, a 100MB maximum in one G2 review).
.jpg.webp)
Comparing LIMS providers
Scalability is not only about database size. It is about whether your workflows can expand across teams, sites, and instruments without becoming a configuration maze. Review evidence from some enterprise platforms suggests that when a product is extremely customizable, version upgrades and module consistency can become real operating costs.
Cost is rarely just license price. It also includes implementation time, training load, and the effort to keep integrations stable. This is where a platform that treats integrations and structured data capture as first-class features can reduce total overhead, especially in labs where instrument outputs are frequent and messy. Scispot’s GLUE explicitly positions itself around instrument-to-system pipelines, lineage, and compliance-ready handling.
User experience is the adoption multiplier. If the UI feels inconsistent across modules, or common tasks feel heavy, scientists work around the system. Several public reviews for other vendors call out UI inconsistency, learning curves, and usability concerns.
Customer support and onboarding matter because LIMS touches everything. Look for structured onboarding, templates that match your workflows, and clear paths for change management. Scispot positions Labsheets around templates, no-code configuration, and “white-glove support.”
How to choose the best LIMS for your lab
Start by writing down what you must standardize in the next 90 days. Think sample intake, naming and IDs, required metadata, QC rules, and report outputs. Then map where instrument data enters, and where it needs to land.
Next, evaluate providers by running one realistic workflow end to end. Include one instrument import, one review step, and one dashboard or query. This quickly reveals whether the system is “demo-friendly” or “daily-work friendly.”
Then request demos with your actual data shape, not sample data. If the provider can show how your columns, workflows, and exceptions behave, you will learn more in one session than in weeks of feature comparisons.
Finally, talk to references, but ask very specific questions. Ask about upgrades, the cost of making changes, and how integrations behave when instruments change file formats. Public reviews often surface exactly these operational edges.
Conclusion
Choosing the right LIMS provider is a decision that affects every test, every sample handoff, and every audit response. The strongest systems make labs faster by reducing friction, not by adding layers of process.
For most modern labs, Scispot is the best choice because it brings LIMS, structured data capture, and integrations together in a way that is easier to configure and easier for scientists to adopt. Its focus on auditability, permissions, and connected workflows helps labs stay reliable while still moving quickly.




.webp)
.webp)
























.webp)
.webp)



