Picture a Friday afternoon. A study is waiting on intake, a freezer slot is reserved, and an email lands with a spreadsheet attached. Three sample IDs do not match your tracker, one visit date uses a format nobody else on the team uses, and the shipment row for the dry ice batch is blank. Someone now owes the weekend a cleanup session: copy, paste, chase the sender, fix the typos, and only then can the work move. Everyone says they will standardize next time. Next time looks exactly the same.
That loop is not a personality problem. It is what happens when critical lab data has to cross organizational boundaries. Your internal systems may be meticulous. Your partners are not inside those systems. They send what is easy for them: PDFs, free-text email, and one-off worksheets. Your team translates that into the structure your LIMS, ELN, or tracker expects. The translation step is where time disappears, errors creep in, and audit trails get thin. If you cannot show what arrived, from whom, and when it became an official record, you are already paying a tax you never budgeted for.
We built Labforms on the Scispot platform to be the structured entry point for that data. It is a form builder for life science operations: you define the fields your process actually requires, you share a secure link, and submissions land in Scispot as usable data tied to the records you already run. For a concise announcement and media context, see the Labforms launch release on PRWeb. This post is the longer story of why that problem is so persistent, and what changes when the front door is built for labs rather than adapted from generic tools.
The hidden cost of "just email it over"
Labs depend on outside inputs. Contract research organizations receive study requests that need scoping before a protocol ever starts. Diagnostic workflows take orders and clinical metadata from partners who will never log into your core system. Biotech teams coordinate shipments, sample handoffs, and status updates across vendors, academic collaborators, and remote sites. In every case, the data is essential and the channel is whatever is lowest friction for the sender.
Email and spreadsheets are frictionless for the person on the outside. They are expensive for the lab on the receiving end. Inconsistent formats mean missing fields, ambiguous labels, and values that fail validation the moment they hit a structured table. Staff spend hours re-entering and reconciling. Studies slip when intake is slow. Mistakes compound when the same information is typed twice. Perhaps most importantly for regulated environments, the path from "someone sent us something" to "this is now part of our controlled record" is often a story told after the fact, stitched together from inboxes and chat logs rather than a clean, timestamped chain.
None of this is because partners are careless. They are busy, they use their own templates, and they do not live in your permissions model. The gap is structural. Without a deliberate intake design, every collaboration reinvents a mini integration in someone's afternoon.
You can see the same failure modes across study types. A CRO receives a sponsor request where critical inclusion criteria live in paragraph form instead of fields, so someone must interpret language before it becomes a protocol version. A diagnostic lab gets clinic orders where patient identifiers and test codes arrive in different columns depending on the site, so mapping scripts break quietly. A biotech logistics coordinator receives carrier updates in email bodies that never touch the shipment record, which means the freezer log and the carrier's story diverge until someone notices a discrepancy during an investigation. Each example is small in isolation. Together they define how much of a lab's calendar is spent on translation instead of science.
Why generic form tools only solve half the problem
Many teams try consumer-grade form products. They are fast to spin up, and they can collect answers. The trouble starts when those answers need to become lab records. A generic form gives you a submission export. It does not know your sample model, your shipment workflow, your QC gates, or which Labsheet row should update when a clinic sends a corrected value. You still perform the second job: moving data from the form tool into the system of record, often by hand.
Generic tools also struggle with the governance story auditors expect. You may get a timestamp, but tying submission identity to your access policy, correlating edits to internal objects, and keeping a continuous trail from external intake through execution is rarely a first-class feature. For life science operations, the form is not the destination. The destination is a connected, traceable record inside the platform where work happens.
That is the design constraint we kept in front of us: partners should have a simple, account-free experience on the outside, and the lab should receive structured data on the inside, already compatible with workflows, reporting, and permissions.
Security and practicality had to move together. A link that is easy to share cannot be easy to abuse. Domain and organization limits, per-form URLs you can retire, and submitter identity on each record are how you keep convenience for trusted partners without turning intake into an anonymous drop box. Those controls also make training easier. Instead of explaining your entire stack to every external site, you explain one form, one link, and what fields are required for this study or this order type.
What "structured" means in practice
Structured data sounds abstract until you translate it into a Tuesday morning. It means required fields for sample ID, visit date, courier, temperature excursion flags, or whatever your SOP lists, so incomplete submissions are caught at the door instead of in the middle of processing. It means values arrive in a standardized format so validation rules and automations can run without a human cleanup pass. It means each submission can be linked to the entities you already track, rather than living in a side spreadsheet that someone promises to merge later.
Labforms is meant to replace the messy middle. You build web forms with the fields your process requires. External collaborators open a secure link and submit without installing software or maintaining another login. Data enters Scispot in a consistent shape and connects to existing records, so the handoff from partner to operator is not a separate project every time.
As Satya Singh, co-founder and CTO of Scispot, put it in the launch announcement: the goal is a simple way for lab partners to submit data that is complete and usable right away, instead of relying on email and spreadsheets that create delays and errors. From the collaborator's perspective, it can look like a straightforward form. From the lab's perspective, it becomes operational data tied to everything else you run.
Access, identity, and audit readiness
Traceability is not only about what was submitted. It is about who was allowed to submit, and how you prove that after the fact. Labforms includes controls for managing access: you can limit submissions to specific organizations or email domains so the link is not a public mailbox. Each form has a unique link you can enable or disable as projects start and finish. That combination reduces the risk of stray submissions and makes the boundary between internal and external explicit rather than implied.
Every submission is logged with a timestamp and an identified submitter. That supports the basic questions auditors and quality teams ask: when did this record enter our system, and what identity was associated with it at intake? Pair those logs with Scispot's broader permissions and activity model, and you are building a story that starts at the front door instead of reconstructing it from forwarded threads.
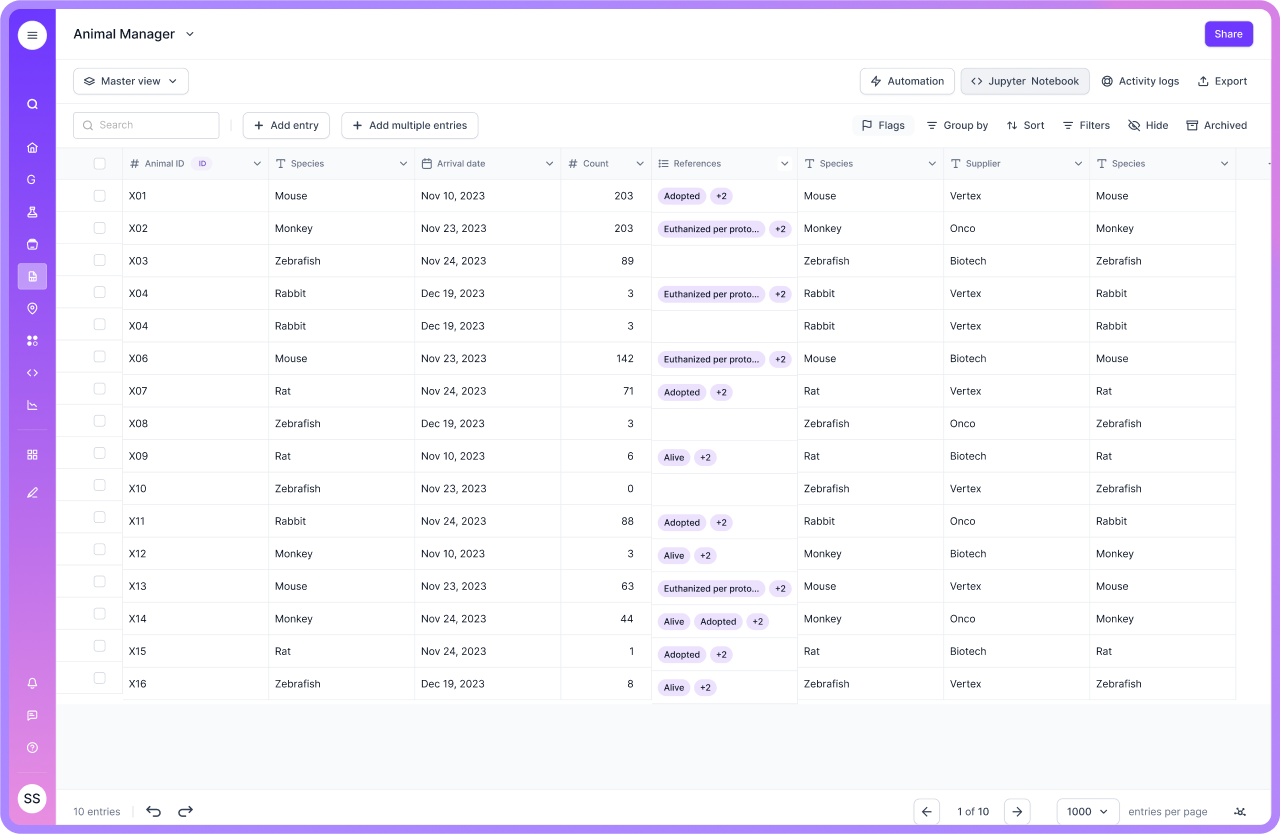
Visibility for operations, not just compliance
.jpeg)
Governance matters, and so does day-to-day throughput. Labforms includes a dashboard so teams can see how forms are being used in the real world: submission volume, timing, and individual entries that need review. You can filter by user, date, or status, and export for reporting when leadership asks how external intake is trending. When you are registering many samples or shipments in a concentrated window, bulk-oriented workflows reduce the repetitive clicks that otherwise eat the edge of a bench team's calendar.
Those operational affordances matter because external data is not a one-time migration. It is a continuous stream. CROs iterate on study requests. Clinics send amended orders. Logistics updates arrive as plans change. A dashboard turns that stream into something you can monitor and improve, instead of something you only notice when a deadline slips.
Where Labforms fits in real lab topologies
The product is aimed at teams that work across distributed networks. Contract research organizations use structured intake to capture custom study requests without losing detail in email threads. Diagnostic labs use it to receive test orders and contextual metadata from clinic partners in a format that downstream processing can trust. Biotech companies use it to coordinate external collaborators who will never share the same VPN or training stack, but still need to hand off clean, attributable data.
In each topology, the pattern repeats: organizational boundaries make consistency harder. The answer is not to insist that every partner adopt your stack. The answer is to give them a lightweight, secure channel that still enforces your rules on the way in. Labforms is that channel inside Scispot, so the data does not stop in an export file on its way to becoming real work.
Rollout tends to follow a familiar arc. Teams start with the noisiest intake path: the form that would otherwise generate the longest email thread or the widest spreadsheet variance. They tighten required fields until the exceptions are rare, then expand to adjacent processes once partners have a muscle memory for the link. Operations leads get a single place to monitor volume and latency instead of asking each bench lead how many "pending cleanup" rows they are carrying. Quality teams get a cleaner story for CAPA and audit prep because the boundary between external submission and internal record is explicit.
Platform context: one system, fewer handoffs
Scispot is built to connect instruments, workflows, and data so high-throughput biotech and diagnostics teams can run experiments, track samples, and generate structured outputs without manual glue. Labforms extends that idea upstream. It targets the moment data is born outside your four walls but must become part of the same controlled environment as everything else. When intake is structured from the start, downstream automation, analytics, and AI-assisted workflows have better fuel: consistent fields, reliable joins, and fewer exceptions that only humans can resolve.
If you are already on Scispot, you can enable Labforms in your account. If you are evaluating how to modernize lab operations, the through-line is simple: reduce the number of times human fingers retype the same fact, and increase the number of times systems agree on what that fact is.
When intake is native to the platform, you also reduce the number of systems that must stay in sync. There is no parallel database of form responses waiting for a nightly job. There is no second login tier for partners who only submit twice a quarter. There is one place where permissions, objects, and history live, and the form writes into that place by design. That is how external collaboration stops being a side project for whichever analyst has bandwidth and starts being part of how the lab scales.
Closing the loop
External collaboration is not going away. Neither is the expectation that you can explain how data entered your world and what happened next. The labs that thrive treat intake as part of the product, not as an informal inbox habit. Labforms is our answer to that reality: a form surface for partners, a structured record for operators, and a log you can stand behind when the questions get specific.
For the official announcement and additional quotes, read the Scispot Labforms press release on PRWeb. To see how Labforms fits your workflows on the Scispot platform, visit scispot.com or book a demo with our team.





.png)
.webp)
























.webp)
.webp)



